Supply Chain Guide to Spinal Biologics
Healthcare supply chain professionals often encounter categories of products that appear somewhat mysterious and difficult to understand. These products can be quite complex and are often employed in intensive procedures and surgeries, and pose unique challenges for supply chain and value analysis efforts.
Biologics are one such product category that is composed of many different technologies and sold by hundreds of suppliers. The purpose of this article is to provide a guide to understanding the biologics that are used in spine surgeries. Understanding the different categories, types of products, and the procedures in which they are used will give you a framework for use when discussing these products with your clinicians and value analysis teams.
How Biologics Are Used
Although biologics have various clinical applications such as trauma and foot and ankle, this article will focus on the biologics used in spinal fusion surgeries.
Spinal fusion is a surgical procedure used to correct problems with the spine by fusing together two or more vertebrae so that they heal into a single, solid bone. The biologics, commonly referred to as bone graft substitutes, are filled into the space between the vertebrae and often inside hardware if used, to promote the growth ofnew bone and stimulate fusion of the vertebrae. For example, the image below shows a pre-op MRI and a post-op CT scan of a patient who had spinal cord compression and severe myelopathy treated with a C4-C5 corpectomy, which resulted in complete bony fusion. The procedure utilized a cage that incorporated two different types of biologics – a premium allograft (OSTEOAMP®) and autograft (patient’s own bone).
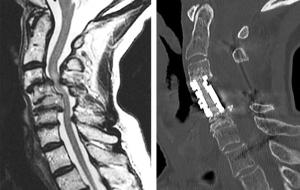
Patient: 68-year-old female
Procedure: C4-C5 corpectomy
Biologics: OSTEOAMP sponge and autograft
Outcome: Pre-op MRI (left), 16 month post-op CT (right) confirms fusion
Type of Biologics
The types of biologics and bone graft substitutes used in spine fusion procedures can significantly affect the overall cost, so understanding this category is useful when looking for opportunities to reduce supply spend or with value analysis considerations. Bone grafts are typically organized into 4 categories: autograft, allograft, growth factor/peptide, and synthetic. Excluding synthetics, most are produced using human bone.
1. Autograft
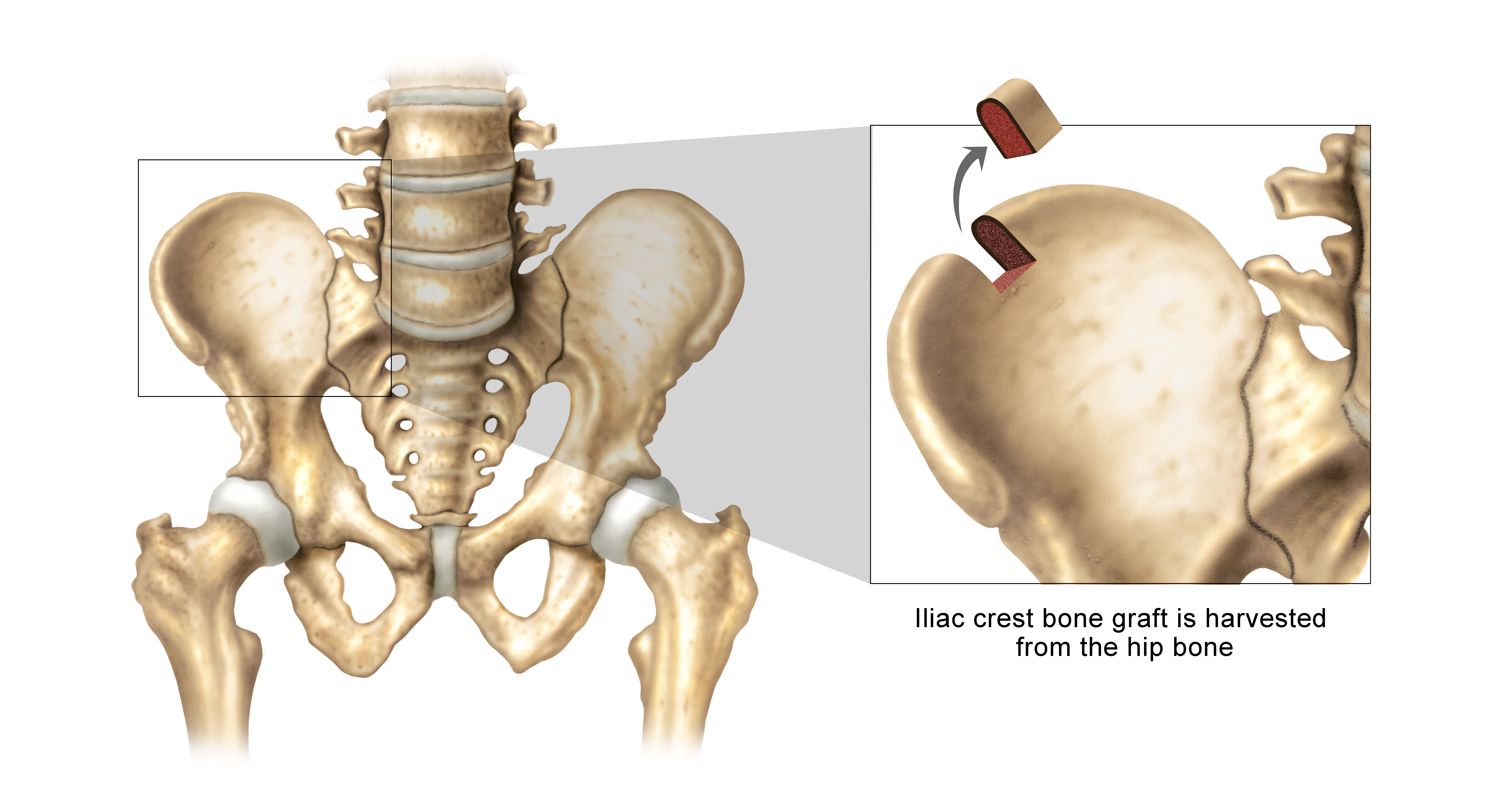
Autograft is bone, tissue, or cells harvested from one site of the patient and transplanted to another site in the same patient. Autologous bone is considered the gold standard of bone grafts as it possesses osteoconductive, osteoinductive, and osteogenic properties. While autologous bone can be collected from multiple locations, including the site of implantation, a primary source of autologous bone is iliac crest bone graft (ICBG) from the hip bone. However, collecting ICBG has several limitations, including potential donor site morbidity following harvesting, and limited supply. Another type of autograft is bone marrow aspirate (BMA). Many surgeons harvest BMA from the patient because it contains osteopro-genitor cells vital to bone healing, and the process to harvest BMA does not have the same donor site morbidity limitations as harvesting ICBG.
2. Allograft
The four types of allograft are listed below:
 Stem Cell Allografts contains stem cells, typically mesenchymal stem cells (MSCs). Stem cells are undifferentiated cells that have the potential of regeneration and differentiation into different types of cells.* Although research is currently inconclusive, use is based on the knowledge of how stem cells operate in the body.
Stem Cell Allografts contains stem cells, typically mesenchymal stem cells (MSCs). Stem cells are undifferentiated cells that have the potential of regeneration and differentiation into different types of cells.* Although research is currently inconclusive, use is based on the knowledge of how stem cells operate in the body. Premium Allografts, such as OSTEOAMP, are uniquely processed with bone and bone marrow to retain naturally occurring growth factors that are present at each phase of the natural bone healing process.1* This unique process yields a product that closely resembles autograft and can be used as an alternative to stem cell allografts, growth factors or peptides.
Premium Allografts, such as OSTEOAMP, are uniquely processed with bone and bone marrow to retain naturally occurring growth factors that are present at each phase of the natural bone healing process.1* This unique process yields a product that closely resembles autograft and can be used as an alternative to stem cell allografts, growth factors or peptides. Demineralized Bone Matrix (DBM) is a form of processed allograft that uses demineralization to expose growth factors found in the bone.24* However, DBM products strip away the bone marrow which also contains growth factors. Additionally, the osteoinductive potential of DBMs can be affected by the specific demineralization process utilized and some DBMs are combined with a carrier for improved handling properties.
Demineralized Bone Matrix (DBM) is a form of processed allograft that uses demineralization to expose growth factors found in the bone.24* However, DBM products strip away the bone marrow which also contains growth factors. Additionally, the osteoinductive potential of DBMs can be affected by the specific demineralization process utilized and some DBMs are combined with a carrier for improved handling properties. Traditional Allografts include mineralized bone chips or powders, as well as structural allografts such as blocks or spacers which can be used in place of hardware to actively support the spine until fusion occurs.
Traditional Allografts include mineralized bone chips or powders, as well as structural allografts such as blocks or spacers which can be used in place of hardware to actively support the spine until fusion occurs.
3. Growth Factor and Peptide
Growth factor and peptide-based bone grafts are manufactured to provide pure concentrations of specific growth factors or peptides via molecular cloning techniques. These products typically provide growth factors or peptides at supra physiological levels. One of the most popular growth factor-based bone grafts, InfuseTM, has been shown to be effective, but there have been concerns regarding patient safety and cost which has triggered hospitals across the country to look for alternatives.
4. Synthetic
Synthetic bone graft substitutes are an alternative to the allograft-based options listed above, with wide availability, comparatively low cost, and absence of risks such as donor site morbidity and transmission of human viruses. Synthetic bone graft substitutes come in a variety forms, including putty, granules, and strips. Synthetic bone graft substitutes can have exceptional handling properties which can be appealing for surgeons that work with a variety of bony voids and musculo-skeletal defects.
Surgeons, hospitals and Integrated Delivery Networks (IDNs) all have unique preferences when it comes to biologics. Additionally, the various needs of the patient, the procedure and the facility can require multiple biologics formats and products.
Understanding the types of biologics used in spine surgeries will be useful when discussing these products with your clinicians and value analysis teams. We hope this article has given you a framework to use when having those discussions.
Properties of Biologics
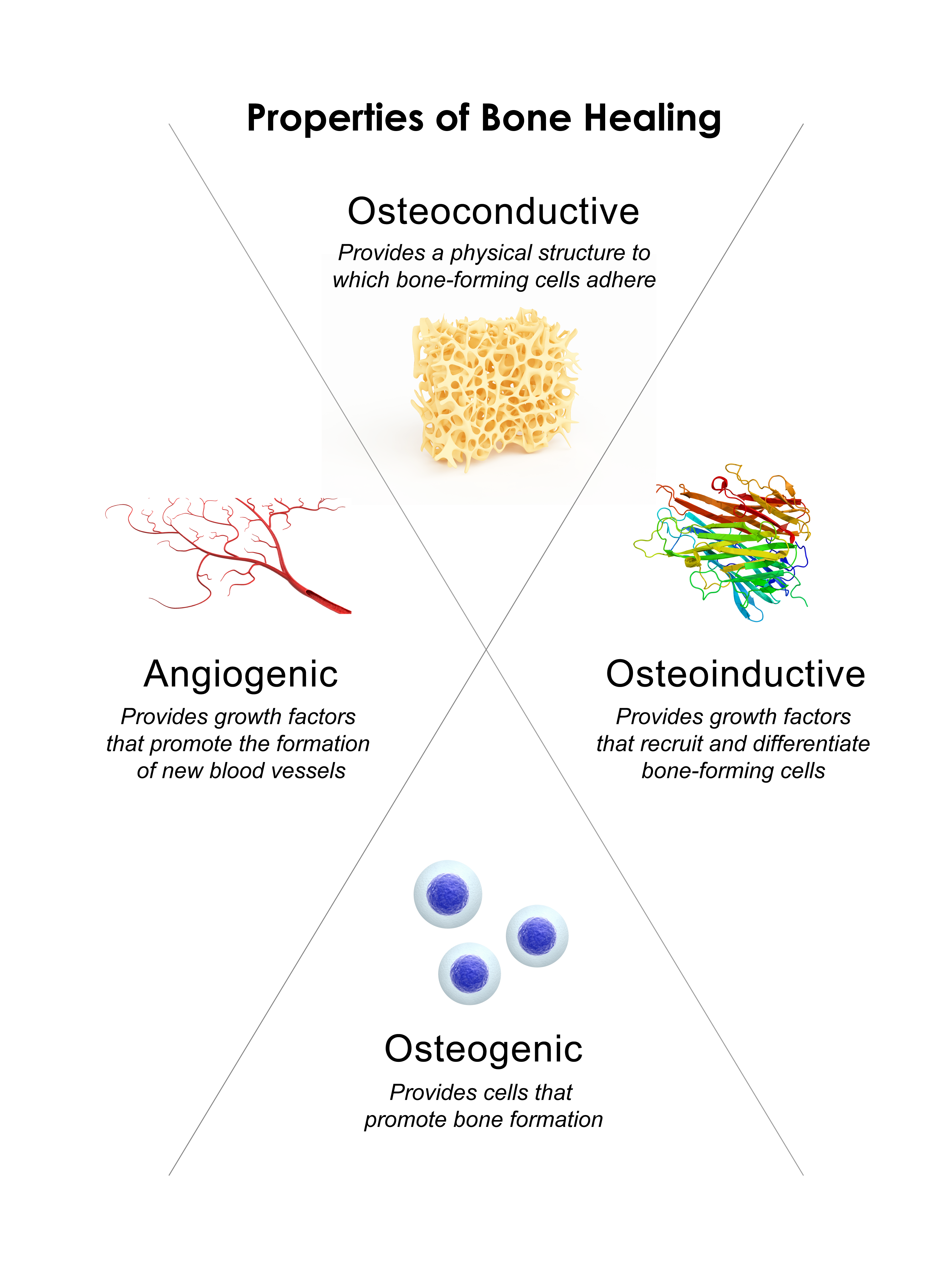
Historically, there are three essential bone healing properties of biologics used in spine surgeries – osteoconductive, osteoinductive, and osteogenic.24 Biologics that are considered osteoconductive contain a physical scaffold or structure to which bone-forming cells adhere to and subsequently build new bone. Osteoinductive biologics contain signaling molecules (growth factors, e.g., BMPs) that help recruit, differentiate, and enable proliferation of the bone-forming cells during the bone healing process. Osteogenic biologics contain the bone-forming cells that support bone healing. More recently, studies have suggested that angiogenesis, the growth of new blood vessels, is increasingly important in the bone healing process by supplying oxygen, nutrients, and precursor bone-forming cells.25 Some biologics claim to be angiogenic in nature. Each of these bone healing properties are inherent to the patient, but can also be provided or supplemented with bone graft substitutes, and a diagram of these properties can be found below.
Please see Instructions for Use for a complete list of indications, contraindications, warnings, and precautions on the product label, by calling 1-800-637-4391 or at www.BioventusSurgical.com.
OSTEOAMP is a minimally manipulated allograft which is compliant to 21 CFR 1271 and is regulated as a 361 HCT/P (ie, human cells, tissues, and cellular and tissue-based products regulated solely under Section 361 of PHS Act). OSTEOAMP is not combined with a drug, medical device, or carrier. As a 361 HCT/P, OSTEOAMP does not require prior FDA approval for commercial distribution (ie, no premarket clearance/approval by FDA is required).
OSTEOAMP may be used in situations where an autograft is appropriate. It should be restricted to homologous use for the repair, replacement, or reconstruction of musculoskeletal defects.
Bioventus, the Bioventus logo and OSTEOAMP are registered trademarks of Bioventus LLC.
INFUSE is a registered trademark of Medtronic.
*In vitro performance may not be predictive of performance in humans.